Charles Law Calculator
The Charles Law Calculator is a free online tool that shows how much a gas expands when heated. The online Charles law calculator tool available here speeds up the calculation and displays the gas volume in a fraction of a second.
Charles' Law - Definition
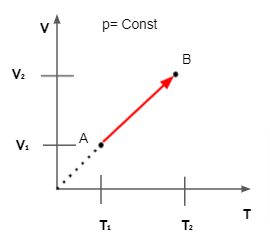
When the pressure and mass of the gas are constant, Charles' law (also known as the law of volumes) describes the relationship among the volume of a gas and its temperature. The volume is proportional to the absolute temperature, according to this formula.
The volume of an ideal gas is precisely proportional to its absolute temperature at constant pressure, according to Charles' Law.
Charle's Law Formula
The volume of gas for a fixed mass is directly proportional to the temperature, according to Charles law in chemistry. It's also known as the volumetric law. This law describes how a gas expands as its temperature rises. The ideal gas law is a specific case of Charles law.
The following is the formula for calculating the volume of a gas under two different conditions: T2/T1 = V2/V1
- Where, the starting and final temperatures are T1 and T2, respectively.
- V1 and V2 are the first and last volumes, respectively.
How to Use Charles Law Calculator?
Charles Law Calculator is pretty straightforward to use. Learn how to use the free online calculator for Charles Law in the below modules. They are as follows
- In the input field, enter the volume, temperature, and x for the unknown.
- To calculate the volume of gas, click the "Calculate x" button.
- Finally, the output field will display the result.
Charles Law Examples
Example 1:
Imagine a ball that has been pumped full of air. It has a starting volume of 2 litres and is placed on a beach with a temperature of 35°C. Then we put it in an air-conditioned chamber with a temperature of 15 degrees Celsius. How does the ball's volume change?
Solution:
First and foremost, Charles' law formula needs absolute temperature readings. Hence we must convert them to Kelvin: T1 = 308.15 K, T2 = 288.15 K, T1 = 35°C = 308.15 K.
We can then use the Charles' law calculation to get the final volume: V2 = V1 / T1 * T2 = 2 l / 308.15 K * 288.15 K = 1.8702 l.
As can be seen, the volume of the ball reduces as it moves from a warmer to a more relaxed environment.
Example 2:
We heat a container that can be easily extended. It's filled with nitrogen, which comes close to being a perfect gas. At 295 K and room temperature, it has an initial volume of 0.03 ft3. After that, we place it near the heat source and leave it for a time, and its capacity has increased to 0.062 ft3 after a few minutes. Can we predict the temperature of our heater using all of this information?
Solution:
Rewrite T2 = T1 / V1 * V2 = 295 K * 0.03 ft3 / 0.062 ft3 = 609.7 K using the Charles' law formula: T2 = T1 / V1 * V2 = 295 K * 0.03 ft3 / 0.062 ft3 = 609.7 K.
T2 = 336.55°C or T2 = 637.79°F is a more palatable form of the result.
Applications of Charles Law in Real Life
Charles' law can be applied in a variety of situations.Here are a few of the more well-known and exciting examples:
Ballon Ride: At some point in your life, you must have seen a balloon in the sky. Have you ever wondered how it can fly and why it is equipped with a fire or other heating source onboard? The answer is Charles' law!
The volume of air expands when it is heated. As a result, the same amount (mass) of gas takes up more space, resulting in a drop in density. The surrounding air's buoyancy takes care of the rest, and the balloon begins to float. The steering in any given direction is almost certainly different.
Experiments with Liquid Nitrogen: Have you ever seen an investigation in which a ball or balloon is placed inside a liquid nitrogen container and then transferred outside? To begin with, it shrinks no matter how big it was at the start; the unit then returns to its original state after being liberated. When the temperature varies, the volume changes as well.
A thermometer: It is possible to build a device that gauges temperature using Charles' law, as illustrated in the previous section. Even though we must be conscious of its restrictions, primarily the object's tensile strength and resistance to high temperatures, we can create an original gadget that exactly suits our needs.
FAQ’s on Charles Law Calculator
1. What is the formula for calculating Charles Law?
We can construct the Charles' law equation in the following form based on the definition of Charles' law: V1 / T1 = V2 / T2, where V1 and T1 are the volume and temperature of the initial state, respectively. V2 and T2 are the final values of these gas parameters, respectively.
2. What is the final temperature formula?
To calculate the final heat of your substance, multiply the temperature change by the original temperature. If your water was 24 degrees Celsius before you started, the end temperature would be 24 + 6, or 30 degrees Celsius.
3. What is Amontons law, and how does it work?
When the volume of a gas is held constant, Gay-law Lussac's (also known as Amonton's law) asserts that the pressure of a given amount of gas varies directly with the absolute temperature of the gas.
4. What would the final temperature of a 50g/20 combination be?
Assume that water has a specific heat capacity of S. As a result, a mixture of 50g of water at 20°C and 50g of water at 40°C will have a final temperature of 30°C.
